MYONEX NEWS
Myonex featured in ACT: Overcoming Challenges Associated with Ancillary Supply Management
05/24/23This article was first featured in Applied Clinical Trials Magazine.
When performing study start-up activities for clinical supplies, a significant amount of time is spent planning the sourcing, labeling, and distribution of study investigational products (IP). Less attention is directed toward ancillary supplies, but failing to adequately prepare for sourcing and shipping of ancillaries can result in enrollment delays and increase operational costs. Because of the importance of ancillary supplies to support a trial, it is critical to invest the appropriate time into understanding procurement, regulatory requirements, and import limitations.
DEFINING ANCILLARIES
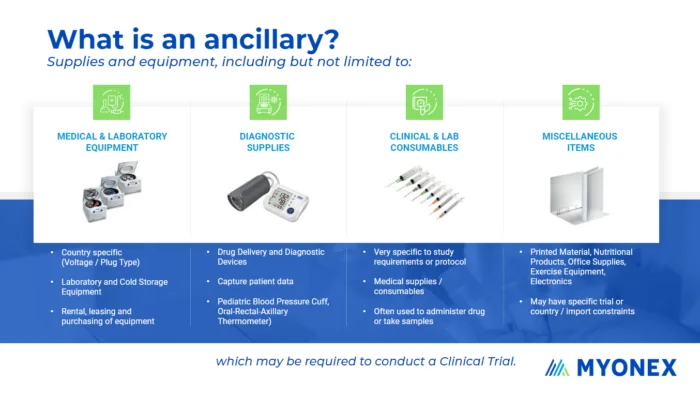
In its most broad sense, an ancillary supply is anything used to support trial operations at the site. An ancillary supply could be something as simple as a notebook or a pen used by a study coordinator or something as large and durable as a refrigerator or a treadmill. As shown in FIGURE 1, an ancillary item most commonly refers to medical and laboratory equipment, diagnostic supplies, clinical site or laboratory consumables, or special storage appliances. The wide range of possible needs makes having a nuanced understanding of supplies and logistics critical for ensuring efficient and timely trial activities.
ANCILLARY MANAGEMENT: COMMON CHALLENGES
The development of a clinical trial requires managing myriad considerations, vendors, and sites, especially for IP planning. Because of the amount of work necessary to ensure the study drug is manufactured and ready for shipping, and the apparent ease of procuring items, ancillary management is often delayed until too late in the planning process. Planning failures can cause significant problems for trial operations. Thus, the protocol stage is the ideal time for beginning ancillary preparations. Although this is before many sponsors start an activity, identifying needs early in the trial planning process can prevent enrollment delays or unanticipated costs. The risks can be illustrated through three case studies based on actual sponsor experiences.
Case Study 1: Unanticipated Supply Chain Impacts
Protocol development and IP planning began in one trial in November 2021, with a planned first Site Initiation Visit (SIV) in November 2022. The delivery of the study drug and placebo required a unique needle gauge. In June 2022, planning began to source the needles.
Component availability impacted supplier production, and in October the sponsor learned the vendor could not provide the needed quantities. Timing could not be worse, as the information was delivered just as shipping was planned to begin. The unavailability of the needles impacted the first SIV, and the ripple effects delayed enrollment and impacted the remainder of the trial.
Case Study 2: Unsuitable Clinical Trial Supplier
In a second scenario, project protocol required sourcing 12 ancillary supplies, 8 of which were available from a common commercial supplier. Because of the anticipated cost savings, the sponsor decided to purchase those items in bulk and ship them from the United States to all 16 countries participating in the study. The third-party vendor contracted to assist with supply logistics identified this as a risk and advised against pursuing this supply strategy, but the sponsor dismissed the concerns.
The items were purchased and kept in stock at the depot, but the shipments were stopped at the country’s borders. When officials requested documentation per country import requirements, the sponsor could not provide the information required due to commercial sourcing.
Case Study 3: Navigating Country-Specific Regulations
In addition to the general import requirements mentioned in the previous case, regulatory agencies have additional requirements for ancillary supplies used in a clinical trial. For example, the European Medical Device Regulation (MDR) covers anything distributed as a medical device in European Union, and the United Kingdom’s similar regulation goes into effect in July 2024.
The sponsor in this next case was performing a diabetes trial in North America, and all supplies were purchased in the United States for domestic and international shipping. Unfortunately, the first shipment was placed on hold when the order arrived at the Canadian border. Canada requires an import license, and any supplies used in a trial must be on the national registry. The lancets purchased were not on the registry, which required pivoting to a new supplier, trial delays, and additional costs.
MITIGATING RISKS WITH ANCILLARY MANAGEMENT
In each case highlighted a lack of planning and expertise significantly impacted study operations, jeopardizing timelines, site relationships, and patient experience. These problems could have been avoided with experienced ancillary management at every stage.
Whether planning to ensure the correct ancillary supplies reach sites before the SIV, understanding the scope to anticipate import regulations, or navigating regulations with supplies and equipment, a third-party supply planning vendor could have mitigated risks.
Kitting Ancillaries
Determining what defines a “kit” can be challenging in the clinical trial space. Because of the wide range of items that can be dispensed as a kit and the possibility of multiple ancillary supplies being combined into a single unit, there is no “standard” kit. When designing kits, carefully considering several concepts can optimize distribution.
Customizing each item adds time and costs to packaging, labeling, and shipping. Rather than individually labeling and dispensing multiple ancillary supplies, consider combining them into a single package, or kit.
Thoughtful Kit Design
Kits can take many shapes and sizes. A kit could be a range of supplies in a clear bag with a single label on the outside. Combining multiple ancillaries in a bag may make kitting easier, but if this packaging could cause damage to the IP, a different design may be warranted. A custom box with sections to hold each included item securely can protect IP from damage.
Individual items may have varying expiration dates, so including them in a kit may require re-packaging when shorter expiry items must be replaced. Consider keeping shorter expiry items separate from the larger kit, then include them in the container at shipping. A detailed reporting tool that shows expiry and shipping lead times can assist in managing kit designs.
Solicit Feedback from End Users
After the kit is designed, ask select sites to provide feedback on kit design, storage considerations, and other best practices. Site feedback may identify shipping container size or storage issues not anticipated in the planning process and can help balance site and shipping needs. Often, sites offer valuable feedback that leads to minor corrections and maximizes site efficiency and collaboration.
Timing and Outsource Kitting Tasks
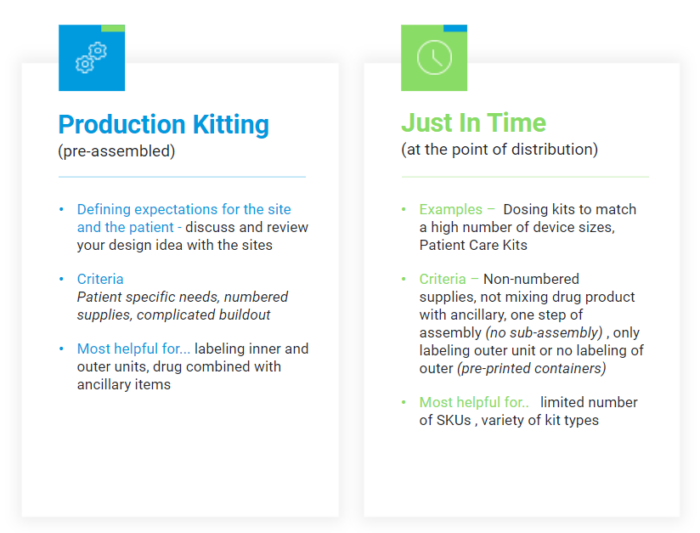
Kitting is best performed based on the components to be included. For more complex or serialized kits, pre-assembly in a production environment is suited to maintain Good Manufacturing Practice (GMP) controls. A Just-in-Time (JIT) approach may provide additional flexibility without sacrificing quality controls for bulk supplies. FIGURE 2 demonstrates several common aspects of each method.
Case Study 4: Pivoting with a Flexible Kit Design
A flexible kit design helped a sponsor pivot when availability issues threatened lead times. The study required 19 ancillary items that the sponsor planned to label individually. Instead, the items were to be combined into a kit which allowed most to be included on a single index form since they were already labeled in their commercial packaging. When supply issues arose, they were easily swapped for available items without label redesign.
Case Study 5: Partnering with a Professional Resource
One sponsor decided to source, pack, and ship ancillary supplies, anticipating it to be easy. However, when the trial began enrolling, the team was quickly overwhelmed by the planning and execution activities required. The team decided to outsource these tasks, which allowed them to focus on core tasks, improved pricing through supplier agreements, and provided better GMP controls.
SELECTING AN APPROPRIATE PARTNER
As illustrated, the anticipated simplicity of ancillary supply management quickly fades once actual planning and shipping activities begin. Working with a partner who specializes in overcoming unusual challenges in ancillary management, from battery shipping regulations to the regional electric appliance variations in Japan, can prevent unnecessary delays and unanticipated costs.
Myonex News

